|
The neon burning process creates additional 16Īre secondary isotopes, meaning their synthesis requires seed nuclei. Is synthesized at the end of the helium fusion process in stars the triple-alpha process creates 12 Has high relative and absolute abundance because it is a principal product of stellar evolution and because it is a primary isotope, meaning it can be made by stars that were initially hydrogen only. Depending on the terrestrial source, the standard atomic weight varies within the range of (the conventional value is 15.999). These days, various factors deplete the ozone layer in the upper atmosphere and the "ozone holes" in the northern and southern polar regions are of particular concern.Natural oxygen is made of three stable isotopes, 16īeing the most abundant (99.762% natural abundance). Ozone is a key component of the upper atmosphere because it has the ability to absorb ultraviolet light in the 220-290 nm range, so preventing harmful rays at those wavelengths reaching the earth's surface. Ozone is present in the atmosphere as well, but at amuch lower level than O 2. Ozone is unstable thermodynamcally with respect to dioxygen but only converts to O 2 slowly in the absence of a catalyst.Īrrangements of ozone molecules, O 3, in the solid state. Ozone condenses to a blue-black liquid (boiling point -111.9☌ and further cooling results in a dark violet-black solid (melting point -192.5☌. 
Ozone molecules are bent with a bond angle of about 117° in the gas phase. 
Ozone, O 3, is a very reactive blue gas characterized by a pungent "electrical" smell (the name ozone derives from the Greek ozein, to smell). It makes up about a fifth of the earth's atmosphere. It is paramagnetic (that is, possesses unpaired electrons) as a result of two unpaired electrons in the two O 2 π* orbitals. Dioxygen, O 2, is a diatomic colourless oxidizing gas but a pale blue liquid upon condensation. Oxygen occurs as two allotropes, dioxygen and trioxygen (ozone). It is essential for respiration of all plants and animals and for most types of combustion. Oxygen is very reactive and oxides of most elements are known. Liquid ozone is bluish-black, and solid ozone is violet-black. Paradoxically, ozone is toxic! Undiluted ozone is bluish in colour. Large holes in the ozone layer are forming over the polar regions and these are increasing in size annually. Aerosols in the atmosphere have a detrimental effect on the ozone layer. 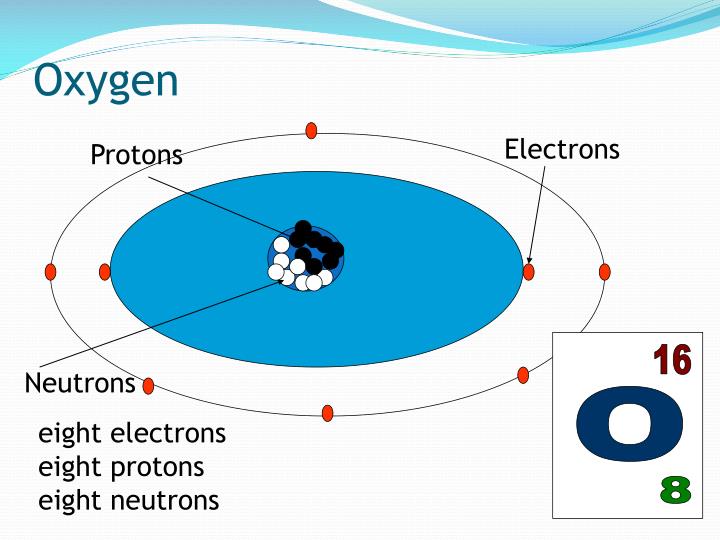
It is an important component of the atmosphere (in total amounting to the equivalent of a layer about 3 mm thick at ordinary pressures and temperatures) which is vital in preventing harmful ultraviolet rays of the sun from reaching the earth's surface. It is formed from electrical discharges or ultraviolet light acting on O 2. Ozone (O 3) is the second allotrope of oxygen. Liquid and solid oxygen are pale blue (see picture above) and strongly paramagnetic (contains unpaired electrons). The gas is colourless, odourless, and tasteless. About two thirds of the human body, and nine tenths of water, is oxygen. Oxygen in excited states is responsible for the bright red and yellow-green colours of the aurora. Oxygen is the third most abundant element found in the sun, and it plays a part in the carbon-nitrogen cycle, one process responsible for stellar energy production. While about one fifth of the atmosphere is oxygen gas, the atmosphere of Mars contains only about 0.15% oxygen.
0 Comments
Leave a Reply. |



 RSS Feed
RSS Feed
